<div class="breadcrumb breadcrumbs"><div class="breadcrumb-trail"> » <a href="https://scienceres-edcp-educ.sites.olt.ubc.ca" title="MSTLTT" rel="home" class="trail-begin">Home</a> <span class="sep">»</span> <a href="https://scienceres-edcp-educ.sites.olt.ubc.ca/secondary/" title="Secondary">Secondary</a> <span class="sep">»</span> <a href="https://scienceres-edcp-educ.sites.olt.ubc.ca/secondary/chemistry/" title="Chemistry">Chemistry</a> <span class="sep">»</span> <a href="https://scienceres-edcp-educ.sites.olt.ubc.ca/secondary/chemistry/atomic-theory/" title="Atomic Theory">Atomic Theory</a> <span class="sep">»</span> Model of the Atom </div></div>
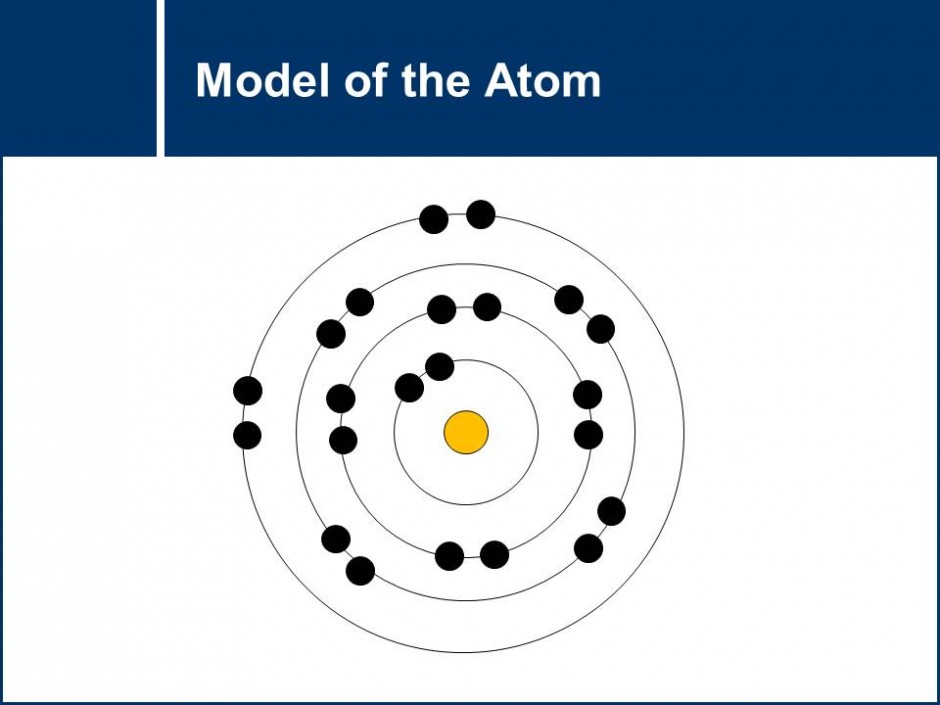
The model of the atom is explored by looking at role and placement of electrons, protons and neutrons in an atom. Questions get students to practice drawing Rutherford-Bohr models of the atom. The role that the gain and loss of electrons have in chemical reactions is also explored which leads to an conclusion about the stability of a full valence electron shell.


Atomic theory, Bohr, Dalton, electron, electron shell, gaining/losing electrons, model of the atom, neutron, proton, Rutherford, stability of full valence shells, Thomson, Thomson-Rutherford model, valence electrons, valence shell
![]()
![]()